Suchitra Ella and Krishna Ella founder of Bharat Biotech got the approval from the Drug Controller General of India for conducting human trials for COVAXIN. Bharat Biotech wiki and biography is also mentioned. Coroflu is also in progress.
Contents
Bharat Biotech COVAXIN Coronavirus vaccine
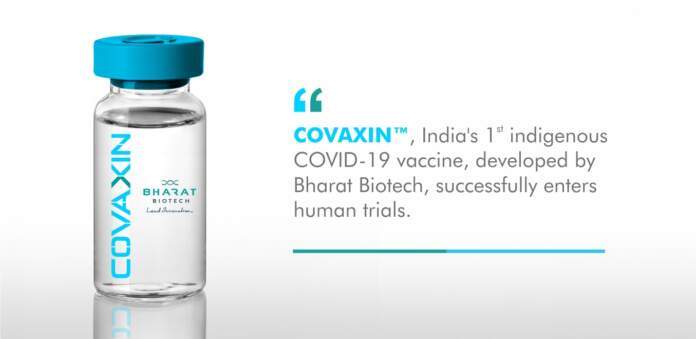
Covaxin is India’s first indigenous inactivated vaccine for highly infectious and deadly disease COVID-19.
The inactivated candidate for the vaccine was produced and manufactured at the BSL-3 (Bio-Safety Level 3 ) high containment facility of Bharat’s Biotech based at Genome Valley, Hyderabad.
See More: Delhi corona city: Delhi becomes worst affected corona city
Co-Founder of Bharat Biotech Suchitra Ella
Mrs. Suchitra Ella is the Joint Managing Director of Bharat Biotech, which she co-founded with Dr. Ella in 1996. With experience in customer operations, finance, marketing and business development, Mrs. Ella is a strong pillar of support and guidance at Bharat, overseeing a wide range of operations in the company.
CMD Krishna Ella
Dr. Krishna Ella is the Chairman & Managing Director of Bharat Biotech International Limited, which he incorporated in 1996. A gold medallist at university, Dr. Ella worked as a research faculty at the Medical University of South Carolina, Charleston after earning his Ph.D. from the University of Wisconsin-Madison.
Bharat Biotech human clinical trials
India, on Monday (June 29) got the approval from the Drug Controller General of India.
for conducting human clinical trials of a potential coronavirus vaccine. The human clinical trials of the country’s ‘first’ indigenous COVID-19 vaccine COVAXIN, which was developed in collaboration with the National Institute of Virology(NIV), IndianCouncil of Medical Research (ICMR) are likely to begin in july.
The Phase I and Phase II clinical trials of the vaccine for SARS-CoV-2, approved after pre-clinical studies demonstrated safety and immune response, would start across the country next month.
Results from these studies have been promising and show extensive safety and effective immune responses, the released added.
Besides Bharat Biotech, at least five other Indian companies are working on a vaccine for the deadly coronavirus while similar efforts are underway in different countries.
Bharat Biotech Wiki, Biography
Bharat biotech wiki and biography: This is not the first landmark for Bharat Biotech as the company had also produced the H1N1 vaccine during the outbreak of swine flu in the country. Reportedly, it has global patents for around 140 products along with a portfolio of over 16 vaccines.
Coroflu is the new vaccine of Bharat Biotech
Apart from Covaxin, Bharat Biotech is partnering with US-based vaccine maker FluGen and virologists at the University of Wisconsin-Madison to develop an intranasal vaccine- coroflu. It has also inked an exclusive deal with the Thomas Jefferson University of Philadelphia for the development of a new vaccine candidate for Covid-19, which has been invented at Jefferson using an existing deactivated rabies vaccine as a vehicle for coronavirus proteins. CoroFlu, is under development for Covid-19 and human clinical trials by Bharat Biotech.
Other companies across the world
Health agencies and pharmaceutical companies across the world are racing against the time to develop an effective vaccine that can curb the spread of the coronavirus, if not eradicate it completely. According to the draft landscape of Covid-19 vaccines by the World Health Organization on June 22, there are 13 experimental vaccines in human clinical trials and other 129 in the pre-clinical evaluation stage.